
Retatrutide 40mg Injection Pen Kit
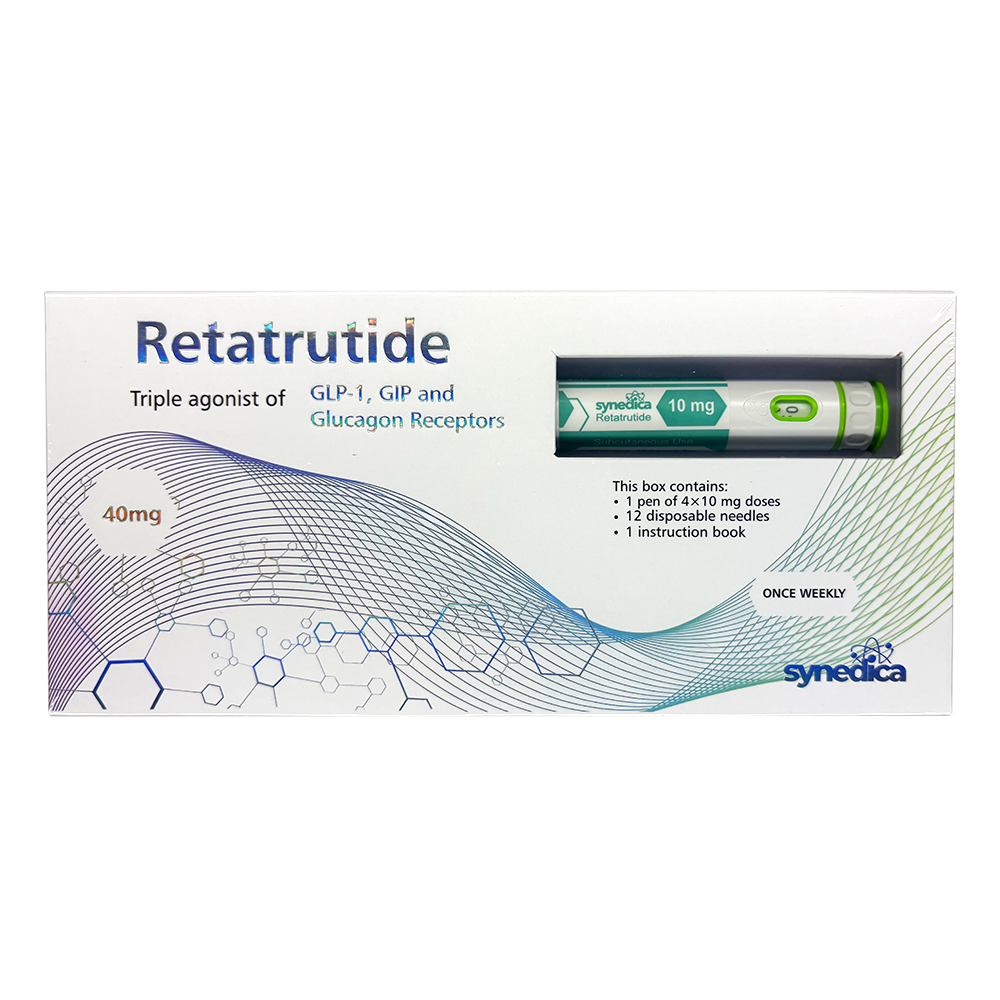


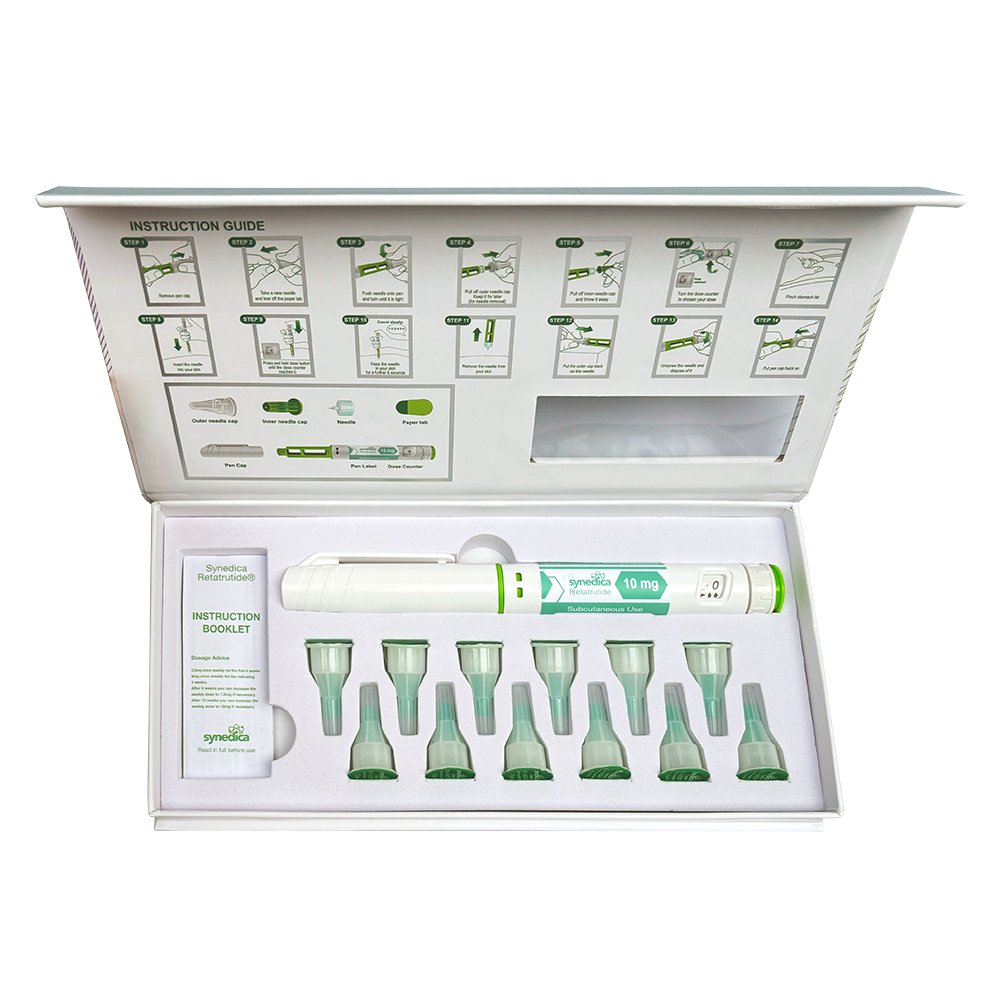
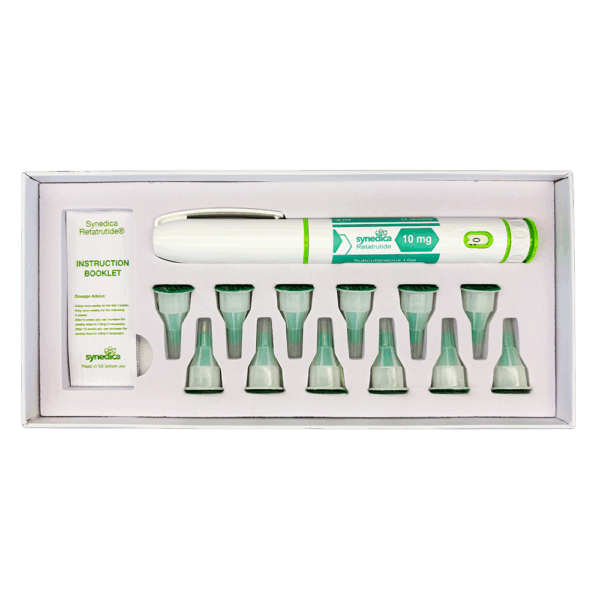


Retatrutide 40mg Injection Pen Kit
Synedica Retatrutide® Injection Pen Kit is the world’s most advanced weight loss solution. Retatrutide is an advanced medication which suppresses appetite leading to significant weight loss. It does this by mimicking the effects of the GLP-1, GIP and glucagon receptors, which are natural hormones that makes humans feel full from eating much smaller portions of food.
Product Details
Synedica Retatrutide® Injection Pen Kit is the world’s most advanced weight loss solution. Retatrutide is an advanced medication which suppresses appetite leading to significant weight loss. It does this by mimicking the effects of the GLP-1, GIP and glucagon receptors, which are natural hormones that makes humans feel full from eating much smaller portions of food.
The Synedica Retatrutide® 40mg Injection Pen Kit contains 1 pen, 12 disposable needles and an instruction booklet.
Each pen has 40mg which can be delivered across 4 full doses of 10mg, or in increments of 2.5mg, 5mg, 7.5mg or 10mg.
Recommended dosage is 2.5mg for the first 4 weeks, then 5mg for the next 4 weeks. After 8 weeks the weekly dosage may be increased to 7.5mg or 10mg if necessary.
⚠️ Important Notice
All products are produced exclusively for controlled laboratory research and development applications.
Not for human or animal consumption.
Suitable R&D Applications
Stability Testing
-
Thermal, enzymatic, and oxidative degradation studies
-
Shelf-life evaluation & formulation robustness
⚗️ Formulation Development
-
Enhances solubility, bioavailability, and structural integrity
-
Tailored for injectable, topical, and research-grade use
Excipient Compatibility Testing
-
Evaluates interactions between peptides & inactive components
-
Ensures safety, stability, and performance
Protease Resistance Evaluation
-
Assesses susceptibility to enzymatic breakdown
-
Supports development of more durable peptide products
Encapsulation & Carrier Systems
-
Liposomes, microspheres, and other delivery vehicles
-
Protects peptides & enables controlled release
Administration Route Assessment
Optimized for:
-
Parenteral delivery (injectables)
-
Dermal absorption (cosmetic/topical)
-
In vitro research applications

 sales@synedica.ltd
sales@synedica.ltd